The electronic and magnetic properties of rare-earth nickelates (RNiO3) are complex and change dramatically with the rare-earth atom (R) and temperature.
PrNiO3 is particularly interesting because its electronic and magnetic changes occur simultaneously and suddenly at the same temperature, making it a great candidate to understand the underlying processes in detail. Such understanding comes from studying single crystals. Prior researchers have only been able to produce extremely small crystals of this material despite using over 40,000 atm pressure.
Using PARADIM’s high-pressure floating zone furnace, a PARADIM user grew mm-size PrNiO3 crystals at pO2=295atm. These crystals are the largest and most perfect of this material produced to date.Analysis of the crystal structure hints that it is different than previously thought.
Hong Zheng et al. Crystals 9 (2019 324.
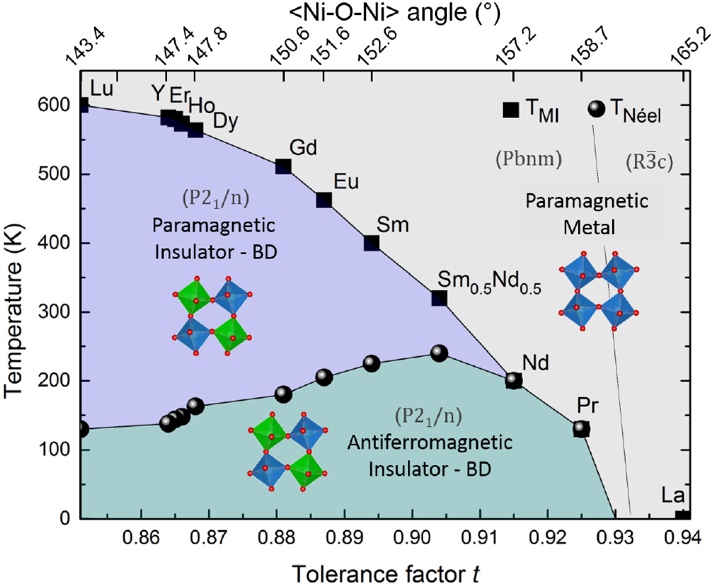
Figure 1: Phase diagram of rare earth nickelates extracted from reference: S Catalano et al., Rep. Prog. Phys. 2018, 81,046501. |

Figure 2: Photograph of the PrNiO3 crystal boule grown under 295 bar of oxygen. |
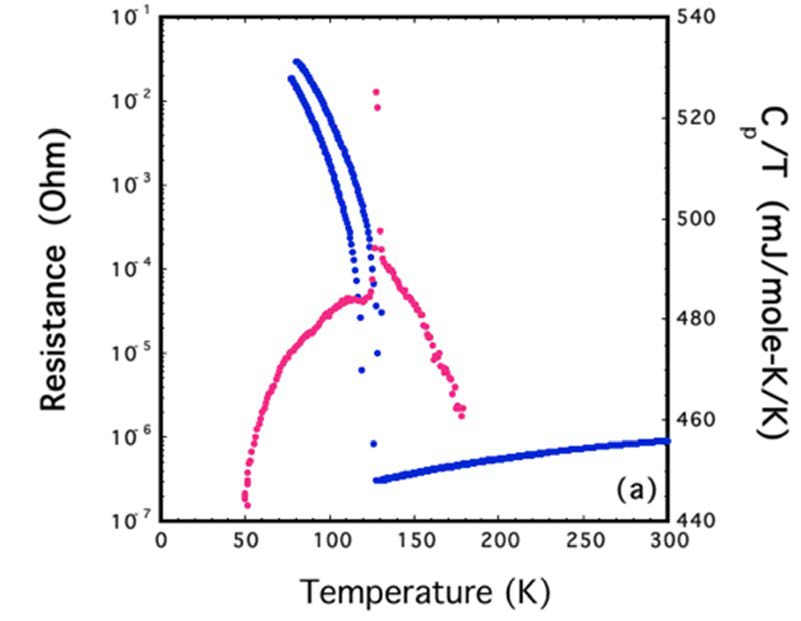
Figure 3: Transport measurement (left) and specific heat (right) of one of the samples. |
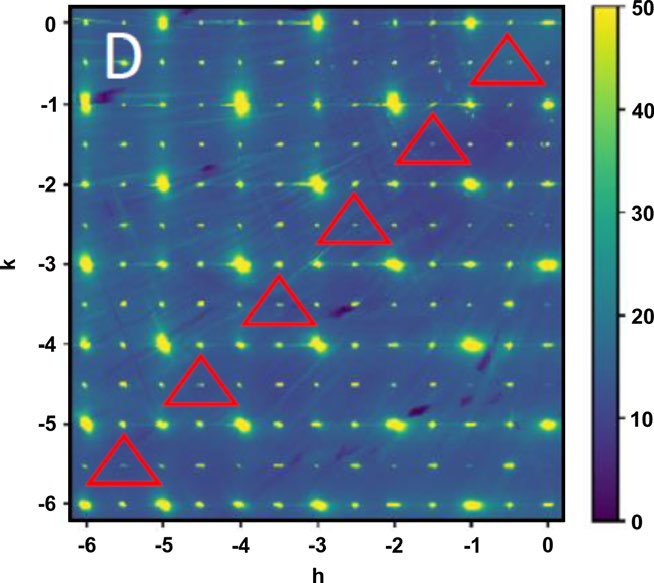
Figure 4: Pseudocubic (hk1) plane measured by single crystal X-ray diffraction at 100K. Triangles indicate positions of points that are forbidden by Pbnm but allowed for P21/n space groups. |
Technical details:
The science and capabilities of crystal growth have historically lagged behind polycrystalline synthesis strategies. With the development of 300 atm floating zone growth, PARADIM is positioned as the only facility in the world capable of utilizing the continuously tunable properties of supercritical fluids – which can dissolve chemicals like a liquid but flows and expands to fill its container like a gas – in the production of high temperature melting functional materials.
The perovskite nickelates, RNiO3 (R = La, Pr-Lu), have received ongoing attention due to their complex magnetic, transport, and structural phase diagram [1]. Except for LaNiO3, all of the others undergo a first-order metal-insulator transition (MIT) as function of temperature. The nature of the MIT has been discussed in the literature in terms of charge disproportionation, negative charge transfer, and bipolaron condensation, but no consensus has been reached. Polycrystalline powders and thin films provide an incomplete means to understand the nature of the structural and magnetic transitions in the perovskite nickelates. Thus, single crystals of size and quality suitable for neutron diffraction are necessary.
It is well-established that high oxygen partial pressure (pO2) is needed to prepare polycrystalline specimens of RNiO3. For single crystal growth, it was reported that single crystals of PrNiO3 with dimensions of ~ 0.5 mm on an edge could be grown by slowly cooling the melt of Pr6O11 + 6NiO + 0.5 KClO4 + 0.5 KCl + 0.5 NaClO4 + 0.5 NaCl at 4.5 GPa by Saito et al. in 2003 [2,3]. Micrometer-sized single crystals of NdNiO3 (~100 µm on an edge) were grown in a belt-type press at 4 GPa by Alonso et al. [4]. Recently, the successful growth of LaNiO3 single crystals using a high-pressure floating zone furnace has been reported, reviving interest in this strongly correlated metallic oxide. The required pO2for stabilizing PrNiO3 is expected to exceed that of the LaNiO3 case, as is true for polycrystalline synthesis of nickelates of the smaller rare-earth ions.
By using the PARADIM 300 bar floating zone furnace, this PARADIM user (John Mitchell) succeed to grow PrNiO3crystals at 295 bar of oxygen pressure. Single crystal x-ray scattering demonstrates the quality of the single crystals and reveals temperature-induced changes near TMIT. Electrical resistance and heat capacity measurements on the grown single crystals evidence MITs and show how the MITs and the low-temperature states respond to oxygen stoichiometry.
Full journal citation:
H. Zheng, J. Zhang, B. Wang, D. Phelan, M.J. Krogstad, Y. Ren, W.A. Phelan, O. Chmaissem, B. Poudel, and J.F. Mitchell, “High pO2 Floating Zone Crystal Growth of the Perovskite Nickelate PrNiO3,” Crystals 9, 324 (2019)
References:
[1] Catalano, S.; Gibert, M.; Fowlie, J.; Íñiguez, J.; Triscone, J.-M.; Kreisel, J. Rare-earth nickelates RNiO3: thin films and heterostructures. Rep. Prog. Phys. 81 2018) 046501.
[2] Saito, T.; Azuma, M.; Nishibori, E.; Takata, M.; Sakata, M.; Nakayama, N.; Arima, T.; Kimura, T.; Urano, C.; Takano, M. Monoclinic distortion in the insulating phase of PrNiO3. Phys. B 329–333 Pt 2 (2003) 866–867.
[3] Azuma, M.; Saito, T.; Ishiwata, S.; Yamada, I.; Kohsaka, Y.; Takagi, H.; Takano, M. Single crystal growth of transition metal oxides at high pressures of several GPa. Phys. C 392–396 Pt 1 (2003) 22–28.
[4] Alonso, J.A.; Muñoz, A.; Largeteau, A.; Demazeau, G. Crystal growth of NdNiO3 perovskite under high oxygen pressure. J. Phys. Condens. Matter 16 (2004) S1277.







